Cover Image: Pan Damman, March 2026
By: Jacob Van Oorschot, Contributing Writer
More uses for mRNA vaccines?
In a talk at the 2024 Trottier Symposium for public science, Nobel laureate Drew Weissman presented work to develop an mRNA vaccine against the pernicious hospital acquired infection C. difficile and mRNA genetic therapy for sickle-cell anemia (1, 2). Weissman also hinted at ongoing work using mRNA vaccines to treat autoimmune diseases, allergies and cancers, as well as pathogens pathogens with no currently approved vaccines, including HIV, Hepatitis C virus, and a lesser-known pathogen called cytomegalovirus (CMV).
By coincidence, the day after Weismann’s talk, Moderna announced it had scrapped a stage III trial for its vaccine against CMV (3–6). CMV is a common virus, to which at least half of the general population have been exposed. Infection is often asymptomatic, but the immune system often fails to produce a response that protects against future infection. This means that reinfection is possible (7). In the occasional cases where CMV does produce symptoms in otherwise-healthy people, the symptoms can resemble those of a cold or flu, or, in worse cases, mononucleosis and hepatitis (similar to its viral relative, Epstein-Barr Virus) (8). Furthermore, CMV can cause worse symptoms, affecting a variety of organs in immunocompromised people (8). And finally. infection during pregnancy carries a high risk of birth defects and developmental issues (7).
The latter issue motivated the Moderna trial, which was conducted in women 16 to 40 years old who had never been infected by CMV before but were at risk of exposure to the virus (9). To tell whether somebody has ever been infected by CMV, a blood sample is tested for antibodies against the virus. If antibodies are present, the patient is “seropositive,” meaning they have been infected at some point in their life. If not, they are considered “seronegative,” meaning they have never contracted the virus (10).
Why did you participate?
To gain more insight into the issue of CMV vaccination as well as this trial specifically, I spoke to one of its participants, who asked to remain anonymous as participants are asked to not identify themselves publicly. She told me that meeting the eligibility conditions was one reason she chose to join the trial “I realized that […] I fell within the target population, and especially once I found out I was seronegative, I’m like ‘Oh well, I It’s not that easy to find people to test this on. It’s a little like being an O-Negative blood donor.” (O-negative is the “universal donor” blood type.) Other factors influenced the decision as well, including “personal curiosity; I’m a scientist, I just thought it would be interesting. I like getting involved in research because I’m interested in other peoples’ research.” She also believed cytomegalovirus to be an important vaccine target.
We take comfort in the idea, in the modern age, that most infectious disease in otherwise healthy adults is preventable or treatable. But due to a lack of demonstrably effective interventions against CMV, the only tools available to pregnant women are behavioural changes to reduce risk of exposure (11). That requires being aware of the risks of CMV in the first place. According to the study participant, “The first time I heard of it [CMV] was when I literally saw a vaccine trial poster in the elevator at the hospital.” Once aware, she was surprised not to have been warned about it sooner.
“There’s really so much lore… there is a general societal thing to tell women not to do things when they’re pregnant that are considerably less risky [than CMV infection]. Like, ‘Oh my God, you ate deli meat? You should have your children taken away from you!’ People are super worried about Listeria, but the chances of getting Listeria when pregnant are actually very small.”
Indeed, congenital cytomegalovirus infection is almost two orders of magnitude more likely than listeriosis during pregnancy, and CMV causes long-term harm to the unborn child more often. Yet awareness of the danger CMV poses, and measures to prevent it, is lower among patients than for listeria (12–14). Listeriosis is a bacterial infection that is usually acquired by eating certain types of raw food. Infection is more likely in pregnant women than in the general population, but still quite rare overall (Listeria infection occurs for about 1 in every 10 000 pregnancies). It is quite dangerous, however, for the unborn child; infection causes death in 20-30% of cases (13). In contrast, congenital CMV infection occurs in at least 1 in every 200 pregnancies, but carries a risk of death below 1% for the unborn child (14, 15). Perhaps the higher mortality risk explains why awareness of listeria is more common than for CMV. While CMV has a large disease burden, it is rarely lethal. This makes it less scary.
Another difference that might explain the difference in awareness is in the type and effectiveness of preventative measures available. Listeria infection is prevented by avoiding certain types of foods (in particular, unpasteurized meat and dairy, but also any raw or prepared food generally), while cytomegalovirus can only be avoided by reducing contact with those at risk of carrying it and basic hygiene practices (12). Modifying contact with others and hygiene practices asks a lot of people, in practice: consider how poor adherence to COVID-19 prevention measures was. People will readily avoid certain foods and specific risky behaviours for a short period. Asking someone to change how they interact with the people around them to minimize the risk of catching CMV can be a tough sell.
Why is there still no approved CMV vaccine?
A lack of urgency could in part explain why there is still no CMV vaccine, but it also might just be a difficult virus to target. CMV has a number of tricks that help it sneak by the immune system, and in turn, make it a difficult virus to vaccinate against. At the end of the day a vaccine is just a way to train your immune system.
As a first example, scientists have found homologs of human immunomodulatory genes in the CMV genome. This means that the virus has stolen our own genes to tell the immune system to calm down, in order to send that message when it is in the course of infecting us (16)!
CMV also hides from the immune system with a strategy called cell-to-cell transmission. Normally, when a virus reproduces, it releases infectious particles from one cell, where they float around until they bump into a new cell they can bind to and infect. In cell-to-cell transmission, virus particles are spread directly from an infected cell to a fresh target cell that is already touching the infected cell. This shields the virus from the watchful eye of the immune system by making some features of the immune response, like antibodies, less effective (17). CMV shares this strategy with the herpes simplex viruses as well as HIV一two other viruses for which we have not yet been able to develop an effective vaccine (18).
Another factor that could explain why CMV is often able to circumvent the immune system is that it has a large genome with relatively high rates of mutation. The consequence of these characteristics is that there is substantial diversity between CMV strains (7), and diversity is a boon for viruses. To generate an effective broadly protective immune response (whether in reaction to natural infection or a vaccine) requires finding a part of the virus that changes little between all these different strains or to use parts from different strains (7). This challenge also presents itself when developing vaccines for other highly diverse viruses, such as HIV and the Hepatitis C virus (19).
Together, CMV’s ability to fool the human immune system, circumvent the human immune system with immunomodulatory genes and cell-to-cell transmission, as well as its extensive diversity of strains, make it difficult for us to mount an effective immune response against it, whether in response to the virus itself or to a vaccine. For many vaccines, generating an immune response on the level of that resulting from natural infection suffices. For CMV, though, that response is not good enough. Since natural infection doesn’t produce complete immunity, the goal of a vaccine is to elicit an immune response far better than that induced by natural infection (7).
Unfortunately, Moderna’s vaccine candidate was not up to the task. Reports say efficacy was lower than had been hoped for (no better than 23%) (3) but my source also commented that:
“The trial coordinator […] said that they were told not enough people had seroconverted for it to be statistically meaningful, like the actual infection rates were lower than they predicted. But all the news releases said that the efficacy was not good. Not that they couldn’t detect the efficacy, but that it was not good, so I don’t know. Those are two seemingly different pieces of information.”
Moderna is continuing another trial with the same vaccine candidate in a different population of people. Aside from pregnant people, another group at risk of complications from CMV infection consists of transplant recipients, because doing so requires immunosuppression and the transplanted organ itself can spread CMV (20). Moderna has a phase II trial ongoing for the use of this vaccine to prevent significant infection for people receiving bone marrow transplants (3).
What vaccine strategies have been tested?
Moderna’s vaccine candidate uses an mRNA platform (Figure 1B), like the Covid-19 vaccine the company developed at the start of the pandemic. It contains instructions for cells to make proteins normally found on CMV’s outer envelope, called gB and PC. Other companies have ongoing trials using more traditional vaccine technologies. For example, GSK (formerly GlaxoSmithKline) is developing a vaccine that is also based on gB and PC but uses a protein subunit platform (Figure 1C) instead of mRNA. With a subunit vaccine, the proteins of interest are already included, ready to stimulate an immune response as soon as they are injected. Finally, Merck is testing one that uses a “disabled infectious single cycle” (DISC) virus, a new type of live-attenuated vaccine (7). This technology produces virus particles that reproduce once in vaccinees’ cells but then are unable to subsequently infect new cells (21) (Figure 1D).
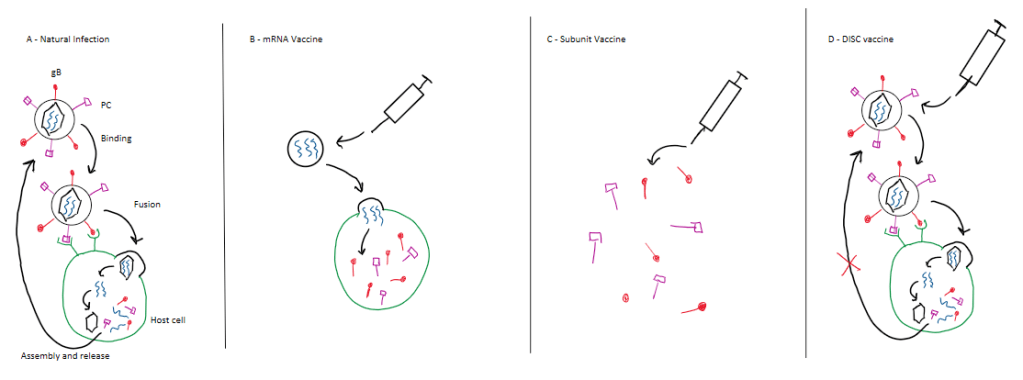
Unfortunately, Merck found an efficacy of only 42% for its candidate in a phase II clinical trial against first-time CMV infection, and it is unclear if Merck will continue its development (7, 22). Past CMV vaccine trials built on the gB protein alone have reached 50% as a high water mark (7). But if that threshold proves hard to cross, getting approval could well mean lowering the bar for efficacy. As long as a vaccine is safe, 50% efficacy is better than the effective 0% rate provided by no vaccine (7). Consider the case of malaria, another disease for which natural infection provides incomplete immunity and vaccine development has proven difficult. In the prevention of malarial disease, at least, it is understood that beggars can’t be choosers; the vaccine used now has an estimated efficacy between 20% and 40%, depending on age (23, 24). The World Health Organization projects that this malaria vaccine could save as many as 500 000 lives by 2035 (24). A CMV vaccine with similar efficacy would no doubt reduce the burden of this tricky virus as well.
The medical research establishment has developed vaccines against many of the most deadly and more tractable diseases, and now has the luxury of turning research efforts towards less feared yet trickier-to-solve diseases including CMV. One hopes these efforts may bear fruit soon.
References
- Weissman D, Schwarcz J. 2025. The Genesis of the COVID Vaccine: The Path to the Nobel Prize.
- Nobel Prize in Physiology or Medicine 2023. NobelPrize.org. https://www.nobelprize.org/prizes/medicine/2023/weissman/facts/. Retrieved 30 December 2025.
- Beaney A. 2025. Moderna scraps mRNA vaccine in congenital CMV on Phase III failure. https://www.clinicaltrialsarena.com/news/moderna-scraps-mrna-vaccine-in-congenital-cmv-on-phase-iii-failure/. Retrieved 8 January 2026.
- Incorvaia D. 2025. Moderna scraps congenital virus program after vax fails ph. 3. https://www.fiercebiotech.com/biotech/moderna-scraps-congenital-virus-program-after-vaccine-shows-little-protective-effect-phase. Retrieved 8 January 2026.
- A Clinical Trial of a Cytomegalovirus (CMV) Vaccine in Healthy Women 16 to 40 Years of Age. Moderna Clinical Trials. https://trials.modernatx.com/study/?id=mRNA-1647-P301. Retrieved 8 January 2026.
- 2025. Moderna, Inc. (NASDAQ:MRNA) | Moderna Announces Phase 3 Study of Investigational Cytomegalovirus (CMV) Vaccine Did Not Meet Primary Efficacy Endpoint. WebDisclosure. https://www.webdisclosure.com/press-release/moderna-inc-nasdaq-mrna-moderna-announces-phase-3-study-of-investigational-cytomegalovirus-cmv-vaccine-did-not-meet-primary-efficacy-endpoint-zDHHhNtIzZx. Retrieved 1 March 2026.
- Permar SR, Schleiss MR, Plotkin SA. 2025. A vaccine against cytomegalovirus: how close are we? J Clin Invest 135.
- CDC. 2025. About Cytomegalovirus. Cytomegalovirus (CMV) and Congenital CMV Infection. https://www.cdc.gov/cytomegalovirus/about/index.html. Retrieved 1 March 2026.
- Trial Details. https://trials.modernatx.com/study/?id=mRNA-1647-P301. Retrieved 1 March 2026.
- Definition of seronegative – NCI Dictionary of Cancer Terms – NCI. nciAppModulePage. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/seronegative. Retrieved 23 January 2026.
- Barton M, Forrester A, McDonald J. Update on congenital cytomegalovirus infection: Prenatal prevention, newborn diagnosis, and management | Canadian Paediatric Society. https://cps.ca/en/documents/position/update-on-congenital-cytomegalovirus-infection-prenatal-prevention-newborn-diagnosis-and-management. Retrieved 22 December 2025.
- Basit I, Crowley D, Geary M, Kirkham C, Dermott RM, Cafferkey M, Sayers G. 2022. Awareness and preventative behaviours regarding toxoplasma, listeria and cytomegalovirus among pregnant women.
- Janakiraman V. 2008. Listeriosis in Pregnancy: Diagnosis, Treatment, and Prevention. Rev Obstet Gynecol 1:179–185.
- Ziebold C, Pillarisetty LS. 2025. Congenital Cytomegalovirus InfectionStatPearls. StatPearls Publishing, Treasure Island (FL).
- Grosse SD, Fleming P, Pesch MH, Rawlinson WD. 2024. Estimates of congenital cytomegalovirus-attributable infant mortality in high-income countries: A review. Reviews in Medical Virology 34:e2502.
- Schönrich G, Abdelaziz MO, Raftery MJ. 2017. Herpesviral capture of immunomodulatory host genes. Virus Genes 53:762–773.
- Murrell I, Bedford C, Ladell K, Miners KL, Price DA, Tomasec P, Wilkinson GWG, Stanton RJ. 2017. The pentameric complex drives immunologically covert cell–cell transmission of wild-type human cytomegalovirus. Proceedings of the National Academy of Sciences 114:6104–6109.
- Zhong P, Agosto LM, Munro JB, Mothes W. 2013. Cell-to-cell transmission of viruses. Current Opinion in Virology 3:44–50.
- Lee J, Woodruff MC, Kim EH, Nam J-H. 2023. Knife’s edge: Balancing immunogenicity and reactogenicity in mRNA vaccines. Exp Mol Med 55:1305–1313.
- Azevedo LS, Pierrotti LC, Abdala E, Costa SF, Strabelli TMV, Campos SV, Ramos JF, Latif AZA, Litvinov N, Maluf NZ, Filho HHC, Pannuti CS, Lopes MH, dos Santos VA, da Cruz Gouveia Linardi C, Yasuda MAS, de Sousa Marques HH. 2015. Cytomegalovirus infection in transplant recipients. Clinics 70:515–523.
- Anderholm KM, Bierle CJ, Schleiss MR. 2016. Cytomegalovirus Vaccines: Current Status and Future Prospects. Drugs 76:1625–1645.
- Das R, Blázquez-Gamero D, Bernstein DI, Gantt S, Bautista O, Beck K, Conlon A, Rosenbloom DIS, Wang D, Ritter M, Arnold B, Annunziato P, Russell KL, V160-002 study group. 2023. Safety, efficacy, and immunogenicity of a replication-defective human cytomegalovirus vaccine, V160, in cytomegalovirus-seronegative women: a double-blind, randomised, placebo-controlled, phase 2b trial. Lancet Infect Dis 23:1383–1394.
- Laurens MB. 2019. RTS,S/AS01 vaccine (MosquirixTM): an overview. Hum Vaccin Immunother 16:480–489.
- RTS Clinical Trials Partnership. 2015. Efficacy and safety of RTS,S/AS01 malaria vaccine with or without a booster dose in infants and children in Africa: final results of a phase 3, individually randomised, controlled trial. Lancet 386:31–45.

