Cover Image: Lilly Guilbeault, January 2026
By: Sagit Yessim, Contributing Writer
If you have ever taken CHEM 211, then chances are you’ve heard the story of Frances Kelsey, a Canadian pharmacologist and a reviewer for the U.S. Food and Drug Administration (FDA) who prevented the import of thalidomide, a drug known for its severe side effects. But how exactly did she do it, and how did thalidomide get onto pharmacy shelves across half the globe?
Frances Oldham Kelsey was born on Vancouver Island in 1914. She earned her B.Sc. from McGill in 1934 and M.Sc. a year later [1]. Due to a lack of job opportunities, Kelsey continued her education abroad, applying for doctoral studies at the University of Chicago. Interestingly, her studies were approved because the professor mistook Kelsey for a male student, “Francis”. However, she proved her abilities, eventually earning a Ph.D. in Pharmacology (1938) and an M.D. (1950) [2].
In 1960, Kelsey took a position as a medical reviewer of new drug applications at the FDA. She was responsible for reviewing the clinical studies and safety data of each new drug. The second application she reviewed was thalidomide.
At the time, thalidomide was a wonder sedative that helped with morning sickness and was widely used by pregnant women. It was already heavily advertised and mass-marketed in 46 countries worldwide before Richardson-Merrell Pharmaceutical Company sought to introduce it to the U.S. [6].
Because Kelsey was still new, her first applications were expected to be easy ones, including thalidomide. Indeed, an affordable and popular sleeping pill – what could go wrong [4]? Yet, upon reviewing clinical studies, Kelsey was surprised to see effectively no safety data; the application contained mere testimonials from doctors complementing the effectiveness of the drug [2]. So, she ruled to reject the application: an obvious decision for her and an unexpected one for the company, too anxious to start selling the drug.
Then, a year-long correspondence began between Merrell and Kelsey, with every new application being repeatedly rejected. During that time, Merrell even attempted to bypass Kelsey and bring the matter to her superiors. They hoped that other FDA personnel would be more convinced by the company’s pompous claims about the drug’s effectiveness and safety [2,6]. At the conference between the FDA and Merrell, for example, the latter argued the drug’s safety by comparing it with barbiturates (then commonly used sleeping pills), saying that thalidomide is safer because “you could not commit suicide with [it]” [2]. The FDA remained unimpressed by these promises about the drug’s safety [6]. Although frustrated with rejected applications, Merrell was still confident in the success of introducing thalidomide, that nearly 2.5 million thalidomide pills were sent out to doctors across the United States for “experimental purposes” [6].
In reality, the company was trying to cover up thalidomide’s toxicity despite growing incidents of alarming side effects across countries [6]. Case reports of nerve damage, drowsiness, dizziness and other adverse effects were ignored and regarded as an agenda against the company [6]. For Kelsey, however, these incidents reaffirmed her decision to reject the application, and she insisted on more thorough clinical studies of the drug.
Merrell’s attempts to push thalidomide to the shelves ended when the drug’s side effects on patients and pregnant women were officially confirmed. Thousands of doctors, mainly in Europe, attributed the cause of patients’ declining health to the use of thalidomide.
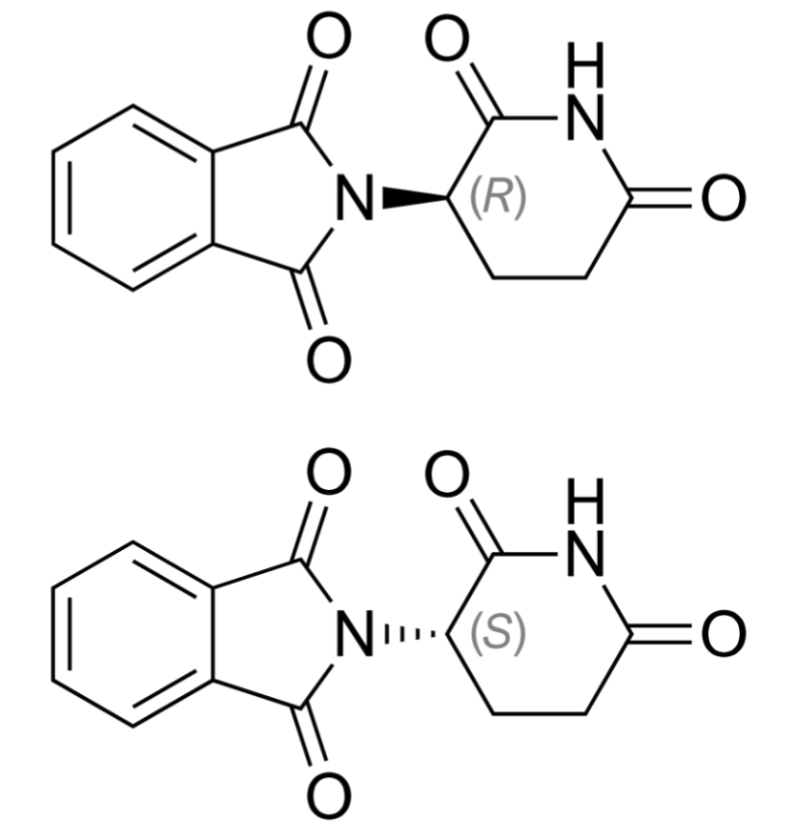
Upon the cessation of thalidomide supply worldwide, 10,000 children had already been born with limb defects, and thousands more patients were diagnosed with nerve damage [5,6]. Were pharmaceutical companies just money-blinded, or was thalidomide’s effect on the human body indeed unpredictable? In the past, the effects of synthesizing thalidomide in a racemic mixture were unknown. Indeed, this complex molecule exists in two states, as shown in Figure 1, with molecules being mirror images of each other (much like our hands). While R-thalidomide is an effective sedative, its mirror copy, S-thalidomide, is responsible for birth defects. Because two molecules can change into one another in our bodies, it’s impossible to isolate them. Due to this structural complexity, thalidomide’s toxicity was hard to notice without thorough investigation.
Despite thalidomide’s complicated and fraught history, it’s now being used for a completely different purpose: to treat cancer. The subsequent rebirth of thalidomide then hints at the following conclusion: the core issue is not a new drug, but how rigorously it is researched and tested. Dr. Kelsey’s vigilance should be a prime example of how scrupulously a drug’s safety should be investigated to prevent needless casualties. Her resistance to corporate pressure also reminds us that private companies should not influence drug regulations. Although the thalidomide tragedy was mitigated in the U.S., the pain inflicted on families in other countries still echoes through generations.
References
- de Bruin, Tabitha. “Frances Oldham Kelsey”. The Canadian Encyclopedia, 12 February 2025. thecanadianencyclopedia.ca/en/article/frances-kelsey
- Kelsey, O. Frances. “Autobiographical reflections.” U.S. Food and Drug Administration. https://www.fda.gov/media/89162/download
- Warsh, C. Krasnick. “Doctoral Studies at the University of Chicago”, Frances Oldham Kelsey, the FDA, and the Battle against Thalidomide. Oxford Academic, New York, 2024, https://doi-org.proxy3.library.mcgill.ca/10.1093/oso/9780197632543.003.0004
- Rehman Waqas, Lisa M. Arfons, and Hillard M. Lazarus. “The rise, fall and subsequent triumph of thalidomide: lessons learned in drug development”. Therapeutic Advances in Hematology. 2011;2(5):291-308. https://doi.org/10.1177/2040620711413165
- James H. Kim, and Anthony R. Scialli. “Thalidomide: The Tragedy of Birth Defects and the Effective Treatment of Disease”, Toxicological Sciences, Volume 122, Issue 1, July 2011, Pages 1–6, https://doi-org.proxy3.library.mcgill.ca/10.1093/toxsci/kfr088